A baby formula recall lawsuit demands compensation if your baby was fed Similac powdered infant formula and suffered a bacterial infection. The US Food and Drug Administration recalled certain powdered baby formulas that may be tied to bacterial infections due to Cronobacter sakazakii and Salmonella. Cronobacter sakazakii is a bacteria that causes life-threatening infections or inflammation of the membranes that protect the spine and brain.
The FDA is currently investigating complaints of at least four infant illnesses across three states, and is advising consumers to not use Similac, Alimentum or EleCare powdered infant formula manufactured at the Sturgis, Michigan Abbott Nutrition facility.
If your infant was fed Similac, Alimentum or EleCare and later suffered from a bacterial infection, contact our award-winning Drug Lawsuit Source attorneys today for your free case review.
- What Symptoms are Caused by the Similac Formula?
- Why Was Similac Baby Formula Recalled?
- Can I Sue Similac?
- Other Lawsuits Against Similac
- Contact a Similac Recall Lawyers
What Symptoms Does the Similac Formula Cause?
Cronobacter sakazakii is a bacteria that can been found in a variety of dry foods, such as powdered infant formula and skimmed milk powder. According to the CDC, Cronobacter illnesses are rare, but are often lethal for infants and can cause sepsis or severe meningitis.
Signs and symptoms to look for include:
- Excessive crying
- Irritability
- Fever
- Poor feeding, stomach pain
- Vomiting
- Swollen abdomen or bloody stool
- Seizures
- Jaundice
- Grunting when breathing
- Low energy
Cronobacter is diagnosed by laboratory culture. Infants will undergo a full evaluation for sepsis, which includes blood, urine, and cerebrospinal fluid cultures.
Which Similac Formulas are Recalled?
The FDA is advising consumers to not use Similac, Alimentum or EleCare powdered infant formulas if the following criteria is met:
- The first two digits of the code on the container are 22 through 37
- The code also contains K8, SH or Z2
- The expiration date is April 1, 2022 or later
On February 28, 2022, Abbott recalled a lot of Similac PM 60/40 also manufactured in Strugis, Michigan. Lot #27032K80, in a can, and Lot # 27032K800, in a case.
Abbott Nutrition said in a statement that they found evidence of Cronobacter sakazakii in their Sturgis, Michigan facility but currently have not found any evidence of the bacteria in products produced in other facilities.
Baby Formula Statistics
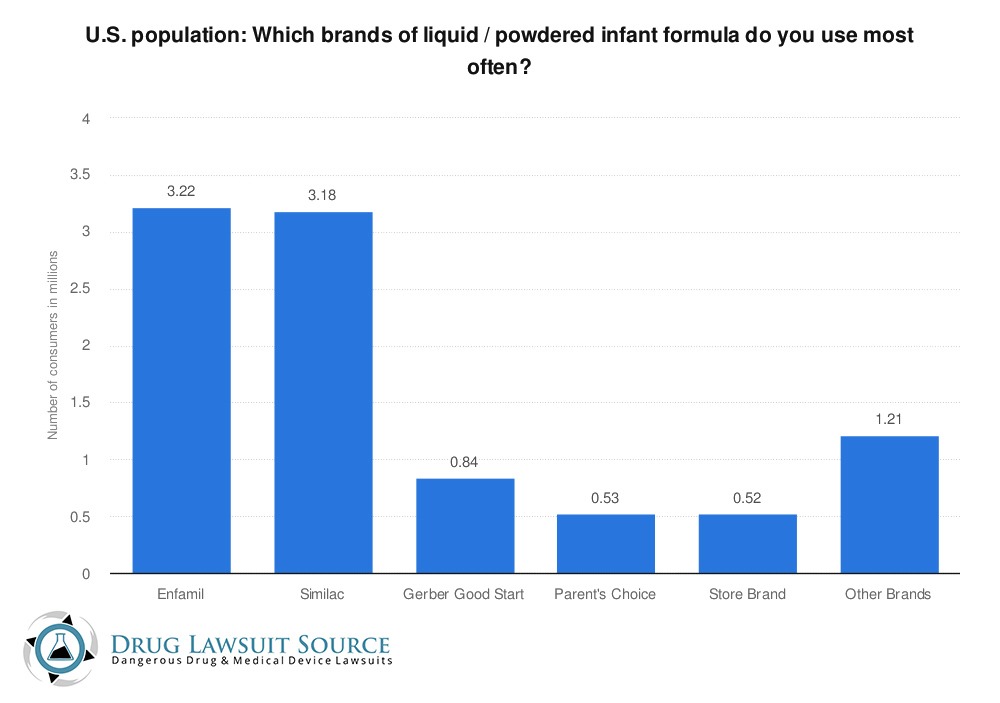
As you can see in the chart below, Similac is the second-most purchased and used brand of baby formula in the United States, according to a survey of US Census data. 3.18 million Americans used Similac formula in 2020.
Can I Sue the Manufacturers of Similac?
To date, no lawsuits have been filed. However, Similac manufacturer Abbott Nutrition is being accused of failing to warn caregivers of the potential bacterial infection risks associated with Cronobacter sakazakii.
According to the CDC, the current manufacturing methods for powdered baby formula do not allow for the possibility to produce sterile powdered formula. It is possible, but not confirmed, that Cronobacter could get into the formula powder if contaminated materials were used to make the formula or if the powder touches a contaminated item in the manufacturing environment.
If your infant developed a bacterial infection due to Cronobacter sakazakii or Salmonella, you may be entitled to compensation.
Additional Lawsuit against Similac
Similac is currently facing other lawsuits filed by caregivers whose infants have been diagnosed with a gastrointestinal illness due to their premature infant being fed cow’s milk-based formula. There are currently no settlements, but litigation has been filed and these claims are being pursued.
Contact Our Baby Formula Lawyers
Contact our award-winning Drug Lawsuit Source attorneys today for your free case review if your premature baby was given Similac and developed a bacterial infection.
Call our baby formula recall lawyers today for your free case review. We charge no fees unless we win. You may be entitled to compensation.
 (866) 280-4722
(866) 280-4722 

